The fate of carbonate in oceanic crust subducted into earth's lower mantle
Drewitt, J. W. E., Walter, M. J., Zhang, H., McMahon, S. C., Edwards, D., Heinen, B. J., Lord, O. T., Anzellini, S., & Kleppe, A. K. (2019). The fate of carbonate in oceanic crust subducted into earth’s lower mantle. Earth and Planetary Science Letters, 511, 213–222. https://doi.org/10.1016/j.epsl.2019.01.041
Carbon is essential for habitability at earth’s surface and understanding how it cycles through exterior and interior reservoirs is fundamental for modelling the evolution of carbon through geological time. Past work by members of the Petrology Group indicates that carbonate reduces the solidus of the subducted oceanic crust, promoting the formation of carbonatitic melts in the transition zone (Andrew R Thomson et al., 2016). We have also shown that diamonds from Juina, Brazil and their silicate mineral inclusions crystallised from just such a carbonatitic melt, suggesting that small-degree melts of subducted crust are efficient agents of chemical mass-transfer in the upper mantle and transition zone, and leave behind a chemical imprint of ocean crust that can possibly endure for billions of years (Bulanova et al., 2010; A R Thomson et al., 2014; Walter et al., 2008). More recently, members of the group showed that these same diamonds host inclusions with compositions comprising the entire phase assemblage expected to crystallize from basalt under lower-mantle conditions while the diamond hosts have carbon isotope signatures consistent with surface-derived carbon, indicating that the deep carbon cycle extends into the lower mantle(Walter et al., 2011).
It is clear therefore that some subducted carbonate can reach the lower mantle. The next question we set out to answer was what happens to this material in the lower mantle? To answer that question, we performed laser-heated diamond anvil cell (LHDAC) experiments in the FeO–MgO–SiO2–CO2 (FMSC) and CaO–MgO–SiO2 –CO2 (CMSC) systems at lower mantle conditions, using the state-of-the-art system installed in the Petrology Group laboratories (Figure 1).

Figure 1: A laser heating experiment in progress in the DAC lab
After synthesising our lower mantle mineral assemblages, we took our run products to beamline I15 at the UK synchrotron facility, Diamond Light Source (Figure 2), and analysed them using X-ray diffraction (Figure 3).

Figure 2: Beamline I15 at Diamond Light Source
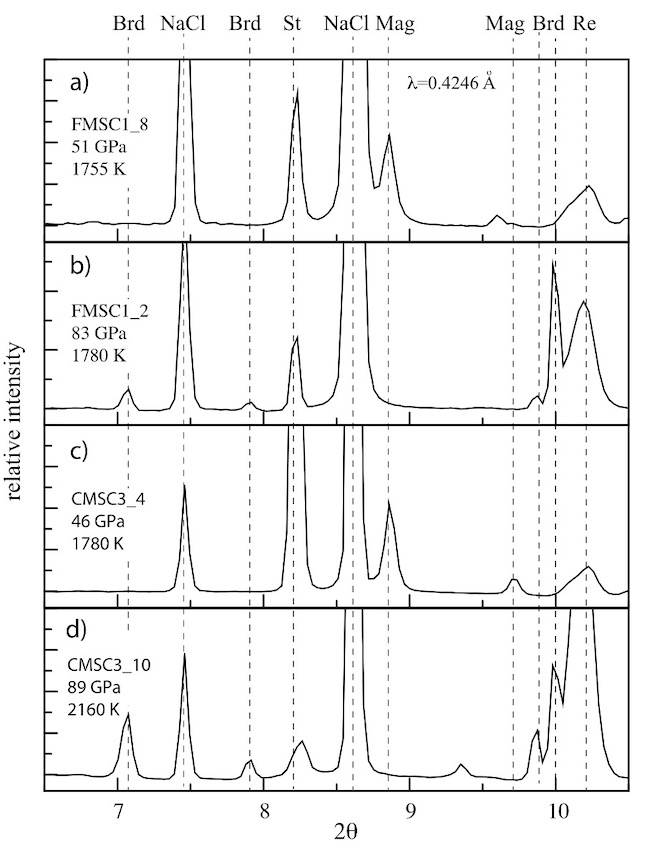
Figure 3: Selected X-ray diffraction patterns showing relative intensity versus scattering angle 2 for P–T quenched run products in the CMSC and FMSC systems. The diffraction lines are labelled according to identified phases; Brd = bridgmanite, St = stishovite; Mag = magnesite as well as the NaCl pressure medium and re gasket.
Our measurements indicate that carbonate phases in these two systems react with silica to form bridgmanite ± Ca-perovskite + CO2 at pressures in the range of ∼40 to 70 GPa and 1600 to 1900 K in decarbonation reactions with negative Clapeyron slopes. Using Raman spectroscopy in the Petrology Group laboratories, we also identified the presence of diamond, due to the dissociation of CO2-V at high temperatures (Figure 4).
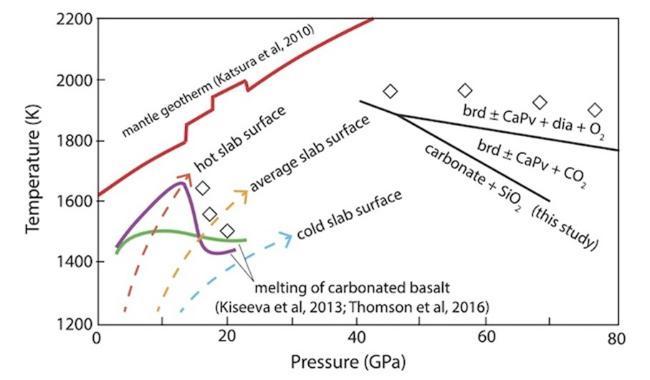
Figure 4: Pressure–temperature diagram showing generalised carbonate phase relations in subducted, carbonated oceanic crust. The melting curves of carbonated basalt at upper mantle and transition zone pressures are from (Kiseeva et al., 2013) (green curve) and (Andrew R Thomson et al., 2016; A.R. Thomson et al., 2016)(purple curve). Also shown are model geotherms at the surface of subducted oceanic crust and a mantle geotherm(Katsura et al., 2010; Syracuse et al., 2010). Diamond symbols show schematically where diamond can be produced via subduction of carbonated oceanic crust.:
These results indicate two important aspects of the deep carbon cycle: that decarbonation reactions form an impenetrable barrier to subduction of carbonate in oceanic crust to depths in the mantle greater than ∼1500 km and that the ultimate fate of carbonate in oceanic crust subducted into the deep lower mantle is in the form of refractory diamond in the deepest lower mantle along a slab geotherm and throughout the lower mantle along a mantle geotherm. Diamond produced in oceanic crust by subsolidus decarbonation is refractory and immobile and can be stored at the base of the mantle over long timescales, potentially returning to the surface in OIB magmas associated with deep mantle plumes.
Other references from the Petrology Group
Bulanova, G. P., Walter, M. J., Smith, C. B., Kohn, S. C., Armstrong, L. S., Blundy, J., & Gobbo, L. (2010). Mineral inclusions in sublithospheric diamonds from Collier 4 kimberlite pipe, Juina, Brazil: subducted protoliths, carbonated melts and primary kimberlite magmatism. Contributions to Mineralogy and Petrology, 160(4), 489–510. https://doi.org/10.1007/s00410-010-0490-6
Thomson, Andrew R, Walter, M. J., Kohn, S. C., & Brooker, R. A. (2016). Slab melting as a barrier to deep carbon subduction. Nature, 529(7584), 76–79. https://doi.org/10.1038/nature16174
Thomson, A.R., Kohn, S. C., Bulanova, G. P., Smith, C. B., Araujo, D., & Walter, M. J. (2016). Trace element composition of silicate inclusions in sub-lithospheric diamonds from the Juina-5 kimberlite: Evidence for diamond growth from slab melts. Lithos, 265, 108–124. https://doi.org/10.1016/j.lithos.2016.08.035
Thomson, A R, Kohn, S. C., Bulanova, G. P., Smith, C. B., Araujo, D., & Walter, M. J. (2014). Origin of sub-lithospheric diamonds from the Juina-5 kimberlite (Brazil): constraints from carbon isotopes and inclusion compositions. Contributions to Mineralogy and Petrology, 168(6), 1081. https://doi.org/10.1007/s00410-014-1081-8
Walter, M. J., Bulanova, G. P., Armstrong, L. S., Keshav, S., Blundy, J. D., Gudfinnsson, G., Lord, O. T., Lennie, A. R., Clark, S. M., Smith, C. B., & Gobbo, L. (2008). Primary carbonatite melt from deeply subducted oceanic crust. Nature, 454(7204), 622–625. https://doi.org/10.1038/nature07132
Walter, M. J., Kohn, S. C., Araujo, D., Bulanova, G. P., Smith, C. B., Gaillou, E., Wang, J., Steele, A., & Shirey, S. B. (2011). Deep Mantle Cycling of Oceanic Crust: Evidence from Diamonds and Their Mineral Inclusions. Science, 334(6052), 54–57. https://doi.org/10.1126/science.1209300
Additional references
Kiseeva, E. S., Litasov, K. D., Yaxley, G. M., Ohtani, E., & Kamenetsky, V. S. (2013). Melting and Phase Relations of Carbonated Eclogite at 9–21 GPa and the Petrogenesis of Alkali-Rich Melts in the Deep Mantle. Journal of Petrology, 54(8), 1555–1583. https://doi.org/10.1093/petrology/egt023
Syracuse, E. M., Keken, P. E. van, & Abers, G. A. (2010). The global range of subduction zone thermal models. Physics of the Earth and Planetary Interiors, 183(1–2), 73–90. https://doi.org/10.1016/j.pepi.2010.02.004
Katsura, T., Yoneda, A., Yamazaki, D., Yoshino, T., & Ito, E. (2010). Adiabatic temperature profile in the mantle. Physics of the Earth and Planetary Interiors, 183(1–2), 212–218. https://doi.org/10.1016/j.pepi.2010.07.001
