
Professor Imre Berger University of Bristol
Without the spike protein, COVID-19 would be unable to enter human cellsUniversity of Bristol

Professor Christiane Berger-Schaffitzel University of Bristol
Press release issued: 13 April 2021
A team of top scientists from the University of Bristol have announced the formation of a new biotech company that is developing ground-breaking and newly patented potential treatments for coronavirus.
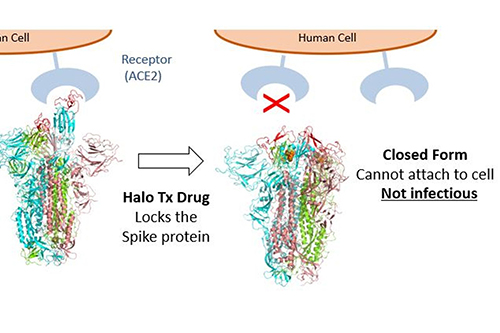
- Halo Therapeutics Ltd is preparing for clinical trials into pivotal, cost-effective antiviral treatments for COVID-19, after discovering a molecule which changes the shape of the virus's spike protein and in so doing inhibits the virus’ ability to enter cells.
- Studies show the treatments are potentially 'pan-corona antivirals' in that they will work against all coronavirus strains - including the highly contagious 'UK (Kent)', 'South African' and 'Brazilian' variants.
- The company is preparing for clinical trials. If approved, the antivirals could be used by patients globally at the first sign of COVID-19 symptoms – stopping the virus in its tracks.
Bristol-based Halo Therapeutics is founded by the team of leading scientists who made a recent breakthrough discovery, which was published in Science. They found that exposing the SARS-CoV-2 (coronavirus) virus to a free fatty acid called linoleic acid locks the virus’s spike protein into a closed, non-infective form stopping it in its tracks.
The company is now preparing to make an application to start clinical trials with infected patients*. If proven to be effective, the antivirals could be used by people of all ages worldwide at the first sign of COVID-19 symptoms, or if they have been in contact with someone with the virus, preventing the virus from taking hold and stopping further transmission.
Lab studies indicate the antiviral will work against all pathogenic coronavirus strains including the highly contagious 'UK (Kent)', 'South African' and 'Brazilian' variants by preventing the virus from penetrating cells in the nose, throat and lungs. The treatments under development by Halo Therapeutics include a nasal spray and an asthma-type inhaler, and offer the possibility of a game-changing pan-coronavirus antiviral to treat patients at all stages of the disease and to reduce the transmission of the virus.
The Halo Therapeutics team is currently engaging investors to help finance multiple parallel clinical trials. If approved, the antiviral treatments could potentially start rolling out to patients globally.
Professor Imre Berger, Director of the Max Planck-Bristol Centre for Minimal Biology at Bristol and one of the team leading the drug’s development, explained: "The aim of our treatment is to significantly reduce the amount of virus that enters the body and to stop it from multiplying. Then, even if people are infected with the virus or exposed to it, they will not become ill because the antiviral prevents the virus from spreading to the lungs and beyond. Importantly, because the viral load will be so low it will likely also stop transmission."
Professor Christiane Berger-Schaffitzel from Bristol's School of Biochemistry added: "Our vision is that at the first sign of the disease, whether you come into contact with someone who has COVID-19 or you have early symptoms, you would self-medicate at home to stop the virus in its tracks and prevent you from getting ill."
Professor Adam Finn from Bristol Medical School and Bristol Vaccine Centre said: "As the virus mutates there is a real risk that presently available vaccines diminish in their protective effect and people could develop the disease again. We need an array of readily-available, cost-effective antiviral treatments that work across all virus strains and complement vaccination efforts."
Further information
The science:
The scientific results, published in the academic journal Science revealed that exposing the SARS-CoV-2 (coronavirus) virus to a free fatty acid called linoleic acid locks the virus’s spike protein into a closed, non-infective form stopping it in its tracks.
While it is known that linoleic acid is indispensable for many cellular functions including inflammation control and maintaining cell membrane elasticity, its central role in COVID-19 has only recently been discovered.
In COVID-19 patients, the body’s limited store of linoleic acid is depleted. This depletion is sensed by an enzyme called cPLA2 which tries to re-supply fatty acid levels in the cell by releasing free fatty acid storage pools from inside the cell. The virus utilizes the released free fatty acids to build specially purposed micro-compartments in which it replicates.
In addition, dysregulated cPLA2 activity causes the membranes of infected cells to stiffen and lose elasticity. In the lungs, this triggers oxygen permeation breakdown that is known to cause breathing difficulties. Ultimately, uncontrolled free fatty acid release contributes proinflammatory precursors which trigger the cytokine storm observed in more severe forms of COVID-19.
Linoleic acid can be purified from plant oils, e.g. sunflower oil, and is scalable to meet global demand. There is a strong potential to create a very cost-effective pan-coronavirus treatment.
The study leading to the discovery included experts from the Max Planck-Bristol Centre, School of Biochemistry, Bristol UNCOVER Group and was supported by funds from the Elizabeth Blackwell Institute with additional support from Oracle high-performance cloud computing and Genscript.
*Phase 2 clinical trial: A phase II clinical trial tells doctors more about how safe the treatment is and how well it works. Doctors also test whether a new treatment works.
What is linoleic acid?
Linoleic acid is a free fatty acid, which is indispensable for many cellular functions. The human body cannot produce linoleic acid: instead, the body absorbs this essential molecule through diet. Intriguingly, linoleic acid plays a vital role in inflammation and immune modulation, which are both key elements of Covid-19 disease progression. Linoleic acid is also needed to maintain cell membranes in the lungs so that we can breathe properly. Linoleic acid is also a central controlling switch for enzymes that release stored free fatty acid from cell membranes.
About Halo Therapeutics Ltd
Bristol-based biotech start-up Halo Therapeutics develops pan-coronavirus antivirals and anti-inflammatory therapies for coronavirus infection and is backed by private investors.
Find us on LinkedIn.
About Professor Christiane Berger-Schaffitzel
Christiane Berger-Schaffitzel is also a Wellcome Trust Investigator and Academic Lead of the Wellcome Trust/BBSRC-funded GW4 Cryo-EM Facility at the University of Bristol, and co-founder and CTO of Halo Therapeutics.
About Professor Imre Berger
Imre Berger is also Director of BrisSynBio, a BBSRC/EPSRC Research Centre for Synthetic Biology in Bristol and a partner in the Wellcome Trust COVID-19 Protein Portal Consortium CPPC. BrisSynBio is part of the Bristol BioDesign Institute. He is co-founder and CSO of Halo Therapeutics.


