Biochemistry under the microscope
Microscope technology is continuously evolving to allow us an ever more detailed look into the workings of biological systems.
rom tissue structure in living organisms to the atomic organisation of protein complexes, the visualisation of biological processes has never been more striking or enlightening.
Explore the way these cutting-edge techniques are applied to the study of biochemistry at the University of Bristol.
Histology
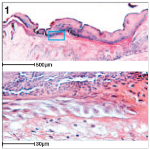 Histology is used to look at the architecture of tissues in organs. Tissue is cut into thin sections and stained so that all the cell layers can be seen. Image 1 is a section showing the organisation of a repairing wound in mouse skin.
Histology is used to look at the architecture of tissues in organs. Tissue is cut into thin sections and stained so that all the cell layers can be seen. Image 1 is a section showing the organisation of a repairing wound in mouse skin.
Confocal Scanning Laser Microscopy
Confocal Scanning Laser Microscopy is used to detect fluorescent molecules that have been added to cells to label specific proteins. Images are created by scanning a laser across the sample to excite the fluorescent molecules, causing them to emit light which is detected by a camera.
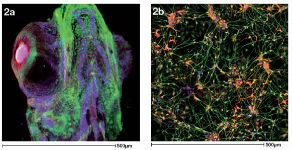 Image 2a is a 3D projection of a zebrafish head in which bones and connective tissues have been labelled in different colours. Image 2b shows a network of neurons grown from stem cells. These images were donated by PhD students Dylan Bergen and Natalia Jimenez-Moreno.
Image 2a is a 3D projection of a zebrafish head in which bones and connective tissues have been labelled in different colours. Image 2b shows a network of neurons grown from stem cells. These images were donated by PhD students Dylan Bergen and Natalia Jimenez-Moreno.
Multiphoton Microscopes
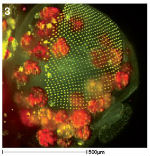 Multiphoton Microscopes are used as an alternative to confocal microscopy for thick samples and live cells. Fluorescent molecules are detected by firing two photons at the sample simultaneously. Only the area where the two photons meet is illuminated and so there is no out of focus light and no photodamage at other sites. Image 3 shows a 3D projection of a fly pupal head with the photoreceptor cells of the eye labelled green and fat cells in red. Image was provided by Anna Franz.
Multiphoton Microscopes are used as an alternative to confocal microscopy for thick samples and live cells. Fluorescent molecules are detected by firing two photons at the sample simultaneously. Only the area where the two photons meet is illuminated and so there is no out of focus light and no photodamage at other sites. Image 3 shows a 3D projection of a fly pupal head with the photoreceptor cells of the eye labelled green and fat cells in red. Image was provided by Anna Franz.
Stimulated Emission Depletion Microscopy (STED)
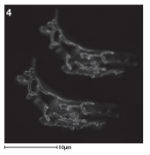 STED is a form of super-resolution light microscopy which functions in a similar way to confocal microscopy except that during laser scanning, fluorescent molecules are selectively deactivated to focus the point of illumination. In image 4 you can see a normal confocal image (top) next to a STED image (bottom) which has much more detail. Image from Janine McCaughey.
STED is a form of super-resolution light microscopy which functions in a similar way to confocal microscopy except that during laser scanning, fluorescent molecules are selectively deactivated to focus the point of illumination. In image 4 you can see a normal confocal image (top) next to a STED image (bottom) which has much more detail. Image from Janine McCaughey.
Scanning Electron Microscopes
 Scanning Electron Microscopes focus a high energy beam of electrons onto a sample. This interacts with atoms on the sample surface to signal back information about its topography (shape) with up to 1nm resolution, allowing scientists to zoom in on tiny structures which cannot be visualised using photons. Image 5a is a scanning electron micrograph of hair-like structures called cilia in the nose of a zebrafish and image 5b is the surface of the lens of a zebrafish eye. Cilia act as antennae to detect odours. Images were provided by Dylan Bergen.
Scanning Electron Microscopes focus a high energy beam of electrons onto a sample. This interacts with atoms on the sample surface to signal back information about its topography (shape) with up to 1nm resolution, allowing scientists to zoom in on tiny structures which cannot be visualised using photons. Image 5a is a scanning electron micrograph of hair-like structures called cilia in the nose of a zebrafish and image 5b is the surface of the lens of a zebrafish eye. Cilia act as antennae to detect odours. Images were provided by Dylan Bergen.
Transmission Electron Microscopy
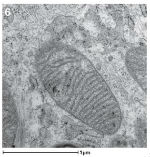 Transmission Electron Microscopy is used to image structures found inside cells and tissues rather than on the surface. Samples are embedded in resin or frozen under high pressure and then sliced into sections that are thin enough to allow electrons to pass through them. Different materials scatter the transmitting electrons differently, creating an image. Image 6 is a section through the middle of a mitochondrion, which provides energy to the cell. Image provided by Nicola Stevenson.
Transmission Electron Microscopy is used to image structures found inside cells and tissues rather than on the surface. Samples are embedded in resin or frozen under high pressure and then sliced into sections that are thin enough to allow electrons to pass through them. Different materials scatter the transmitting electrons differently, creating an image. Image 6 is a section through the middle of a mitochondrion, which provides energy to the cell. Image provided by Nicola Stevenson.
Cryo Electron Microscopy
 Transmission electron microscopy can be performed at cryogenic temperatures to preserve highly delicate or flexible structures. This has become popular in structural biology as it produces sufficient resolution to visualise protein complexes. Image 7 shows electron micrographs of ribosomes sitting in different orientations. These were then used to computationally model the 3D structure of the ribosome as shown on the far right. This image has been artificially coloured. Ribosomes build proteins according to information encoded in RNA molecules.
Transmission electron microscopy can be performed at cryogenic temperatures to preserve highly delicate or flexible structures. This has become popular in structural biology as it produces sufficient resolution to visualise protein complexes. Image 7 shows electron micrographs of ribosomes sitting in different orientations. These were then used to computationally model the 3D structure of the ribosome as shown on the far right. This image has been artificially coloured. Ribosomes build proteins according to information encoded in RNA molecules.
